 | | DRUG: Mexiletine hydrochloride | |
| Entry |
|
|---|
| Name |
Mexiletine hydrochloride (JP18/USP);
Mexitil (TN) |
|---|
| Product |
|
|---|
| Generic |
MEXILETINE HYDROCHLORIDE (ANI Pharmaceuticals), MEXILETINE HYDROCHLORIDE (American Health Packaging), MEXILETINE HYDROCHLORIDE (AvKARE), MEXILETINE HYDROCHLORIDE (Bryant Ranch Prepack), MEXILETINE HYDROCHLORIDE (Bryant Ranch Prepack), MEXILETINE HYDROCHLORIDE (Bryant Ranch Prepack), MEXILETINE HYDROCHLORIDE (Bryant Ranch Prepack), MEXILETINE HYDROCHLORIDE (Bryant Ranch Prepack), MEXILETINE HYDROCHLORIDE (Bryant Ranch Prepack), MEXILETINE HYDROCHLORIDE (Bryant Ranch Prepack), MEXILETINE HYDROCHLORIDE (Bryant Ranch Prepack), MEXILETINE HYDROCHLORIDE (Bryant Ranch Prepack), MEXILETINE HYDROCHLORIDE (Bryant Ranch Prepack), MEXILETINE HYDROCHLORIDE (Bryant Ranch Prepack), MEXILETINE HYDROCHLORIDE (Bryant Ranch Prepack), MEXILETINE HYDROCHLORIDE (Bryant Ranch Prepack), MEXILETINE HYDROCHLORIDE (Camber Pharmaceuticals), MEXILETINE HYDROCHLORIDE (Chartwell RX), MEXILETINE HYDROCHLORIDE (Golden State Medical Supply), MEXILETINE HYDROCHLORIDE (Ingenus Pharmaceuticals), MEXILETINE HYDROCHLORIDE (Lannett Company), MEXILETINE HYDROCHLORIDE (Leading Pharma), MEXILETINE HYDROCHLORIDE (Marlex Pharmaceuticals), MEXILETINE HYDROCHLORIDE (Senores Pharmaceuticals), MEXILETINE HYDROCHLORIDE (Sun Pharmaceutical Industries), MEXILETINE HYDROCHLORIDE (Teva Pharmaceuticals USA) |
|---|
| Formula |
C11H17NO. HCl
|
|---|
| Exact mass |
215.1077
|
|---|
| Mol weight |
215.7197
|
|---|
| Structure |
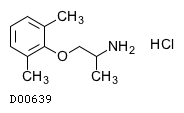
|
|---|
| Simcomp |
|
|---|
| Class |
Cardiovascular agent
DG01653 Antiarrhythmics
DG01652 Class I antiarrhythmic agent
DG01651 Class Ib antiarrhythmic agent
Metabolizing enzyme substrate
DG01892 CYP1A2 substrate
DG01644 CYP2D6 substrate
|
|---|
| Remark |
| Product (DG00197): | D00639<JP/US> |
|
|---|
| Efficacy |
Antiarrhythmic, Antihypertensive, Sodium channel blocker |
|---|
| Target |
|
|---|
| Pathway |
| hsa04261 | Adrenergic signaling in cardiomyocytes |
|
|---|
| Metabolism |
Enzyme: CYP1A2 [HSA: 1544], CYP2D6 [HSA: 1565]
|
|---|
| Interaction |
|
|---|
| Structure map |
|
|---|
| Brite |
Anatomical Therapeutic Chemical (ATC) classification [BR:br08303]
C CARDIOVASCULAR SYSTEM
C01 CARDIAC THERAPY
C01B ANTIARRHYTHMICS, CLASS I AND III
C01BB Antiarrhythmics, class Ib
C01BB02 Mexiletine
D00639 Mexiletine hydrochloride (JP18/USP) <JP/US>
USP drug classification [BR:br08302]
Cardiovascular Agents
Antiarrhythmics
Vaughn Williams-Class I
Mexiletine
D00639 Mexiletine hydrochloride (JP18/USP)
Therapeutic category of drugs in Japan [BR:br08301]
1 Agents affecting nervous system and sensory organs
19 Other agents affecting nervous system and sensory organs
190 Other agents affecting nervous system and sensory organs
1900 Other agents affecting nervous system and sensory organs
D00639 Mexiletine hydrochloride (JP18/USP)
2 Agents affecting individual organs
21 Cardiovascular agents
212 Antiarrhythmic agents
2129 Others
D00639 Mexiletine hydrochloride (JP18/USP)
Drug groups [BR:br08330]
Cardiovascular agent
DG01653 Antiarrhythmics
DG01652 Class I antiarrhythmic agent
DG01651 Class Ib antiarrhythmic agent
DG00197 Mexiletine
D00639 Mexiletine hydrochloride
Metabolizing enzyme substrate
DG01892 CYP1A2 substrate
DG00197 Mexiletine
D00639 Mexiletine hydrochloride
DG01644 CYP2D6 substrate
DG00197 Mexiletine
D00639 Mexiletine hydrochloride
Drug classes [BR:br08332]
Cardiovascular agent
DG01653 Antiarrhythmics
D00639 Mexiletine hydrochloride
Target-based classification of drugs [BR:br08310]
Ion channels
Voltage-gated ion channels
Sodium channels
SCN1A
D00639 Mexiletine hydrochloride (JP18/USP) <JP/US>
SCN2A
D00639 Mexiletine hydrochloride (JP18/USP) <JP/US>
SCN3A
D00639 Mexiletine hydrochloride (JP18/USP) <JP/US>
SCN4A
D00639 Mexiletine hydrochloride (JP18/USP) <JP/US>
SCN5A
D00639 Mexiletine hydrochloride (JP18/USP) <JP/US>
SCN8A
D00639 Mexiletine hydrochloride (JP18/USP) <JP/US>
SCN9A
D00639 Mexiletine hydrochloride (JP18/USP) <JP/US>
SCN10A
D00639 Mexiletine hydrochloride (JP18/USP) <JP/US>
SCN11A
D00639 Mexiletine hydrochloride (JP18/USP) <JP/US>
Drugs listed in the Japanese Pharmacopoeia [BR:br08311]
Chemicals
D00639 Mexiletine hydrochloride
New drug approvals in Europe [br08329.html]
European public assessment reports (EPAR) authorised medicine
D00639
Drug metabolizing enzymes and transporters [br08309.html]
Drug metabolizing enzymes
D00639
Drug groups [BR:br08330]
Cardiovascular agent
DG01653 Antiarrhythmics
DG01652 Class I antiarrhythmic agent
DG01651 Class Ib antiarrhythmic agent
DG00197 Mexiletine
Metabolizing enzyme substrate
DG01892 CYP1A2 substrate
DG00197 Mexiletine
DG01644 CYP2D6 substrate
DG00197 Mexiletine
|
|---|
| Other DBs |
|
|---|
| KCF data |
ATOM 14
1 C8y C 22.8402 -16.9926
2 C8y C 21.6292 -17.7005
3 C8y C 22.8402 -15.6003
4 O2a O 24.0511 -17.6946
5 C8x C 20.4125 -16.9926
6 C1a C 21.6292 -19.0986
7 C8x C 21.6292 -14.8984
8 C1a C 24.0511 -14.8984
9 C1b C 25.2563 -16.9926
10 C8x C 20.4125 -15.6003
11 C1c C 26.4730 -17.6946
12 C1a C 26.4730 -19.0986
13 N1a N 27.6840 -16.9926
14 X Cl 31.3883 -16.8932
BOND 13
1 1 2 2
2 1 3 1
3 1 4 1
4 2 5 1
5 2 6 1
6 3 7 2
7 3 8 1
8 4 9 1
9 5 10 2
10 9 11 1
11 11 12 1
12 11 13 1
13 7 10 1
|
|---|
|
» Japanese version » Back
|
|