| Entry |
|
|---|
| Name |
Emedastine difumarate (JP18/USP);
Daren (TN);
Emadine (TN) |
|---|
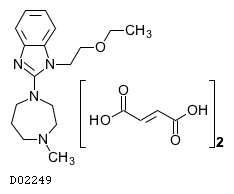
| Formula |
C17H26N4O. (C4H4O4)2
|
|---|
| Exact mass |
534.2326
|
|---|
| Mol weight |
534.5589
|
|---|
| Structure |
|
|---|
| Class |
Anti-allergic agent
DG01557 Histamine receptor antagonist
DG01482 Histamine receptor H1 antagonist
Dermatological agent
DG03243 Atopic dermatitis agent
|
|---|
| Remark |
| Therapeutic category: | 4490 |
| Product (DG01144): | D02249<JP> |
|
|---|
| Efficacy |
Antiallergic, Antiasthmatic, H1 receptor antagonist |
|---|
| Target |
|
|---|
| Pathway |
| hsa04080 | Neuroactive ligand-receptor interaction |
| hsa04750 | Inflammatory mediator regulation of TRP channels |
|
|---|
| Interaction |
|
|---|
| Structure map |
| map07212 | Histamine H1 receptor antagonists |
|
|---|
| Brite |
Anatomical Therapeutic Chemical (ATC) classification [BR:br08303]
S SENSORY ORGANS
S01 OPHTHALMOLOGICALS
S01G DECONGESTANTS AND ANTIALLERGICS
S01GX Other antiallergics
S01GX06 Emedastine
D02249 Emedastine difumarate (JP18/USP) <JP>
Therapeutic category of drugs in Japan [BR:br08301]
4 Agents affecting cellular function
44 Allergic agents
449 Miscellaneous
4490 Miscellaneous
D02249 Emedastine difumarate (JP18/USP)
Classification of Japanese OTC drugs [BR:br08313]
Allergic agents
52 Antihistamine based drugs
D02249 Emedastine difumarate (JP18/USP)
Risk category of Japanese OTC drugs [BR:br08312]
Second-class OTC drugs
Inorganic and organic chemicals
Emedastine
D02249 Emedastine difumarate (JP18/USP)
Drug groups [BR:br08330]
Anti-allergic agent
DG01557 Histamine receptor antagonist
DG01482 Histamine receptor H1 antagonist
DG01144 Emedastine
D02249 Emedastine difumarate
Dermatological agent
DG03243 Atopic dermatitis agent
D02249 Emedastine difumarate
Drug classes [BR:br08332]
Anti-allergic agent
DG01482 Histamine receptor H1 antagonist
D02249 Emedastine difumarate
Dermatological agent
DG03243 Atopic dermatitis agent
D02249 Emedastine difumarate
Target-based classification of drugs [BR:br08310]
G Protein-coupled receptors
Rhodopsin family
Histamine
HRH1
D02249 Emedastine difumarate (JP18/USP) <JP>
Drugs listed in the Japanese Pharmacopoeia [BR:br08311]
Chemicals
D02249 Emedastine difumarate
D02249 Emedastine difumarate extended-release capsules
New drug approvals in Europe [br08329.html]
European public assessment reports (EPAR) authorised medicine
D02249
Rx-to-OTC switch list in Japan [br08314.html]
D02249
Drug groups [BR:br08330]
Anti-allergic agent
DG01557 Histamine receptor antagonist
DG01482 Histamine receptor H1 antagonist
DG01144 Emedastine
|
|---|
| Other DBs |
|
|---|
| KCF data |
ATOM 38
1 C6a C 34.8376 -14.8105
2 C2b C 36.0495 -15.5131
3 O6a O 33.6257 -15.5131
4 O6a O 34.8376 -13.4113
5 C2b C 37.2671 -14.8105
6 C6a C 38.4790 -15.5131
7 O6a O 39.6908 -14.8105
8 O6a O 38.4790 -16.9123
9 C1x C 27.8534 -16.8794
10 C1x C 27.0113 -15.7698
11 C1x C 27.3465 -14.4130
12 N1y N 28.6075 -13.8349
13 N1y N 29.2592 -16.9046
14 C1x C 29.8611 -14.4572
15 C1x C 30.1476 -15.8259
16 C1a C 29.9373 -18.1294
17 C8y C 28.6292 -12.4600
18 N4y N 29.7752 -11.6547
19 C8y C 29.3635 -10.3160
20 C8y C 27.9630 -10.2939
21 N5x N 27.5093 -11.6189
22 C8x C 30.0825 -9.1147
23 C8x C 29.4018 -7.8214
24 C8x C 28.0013 -7.8693
25 C8x C 27.2823 -9.0705
26 C1b C 31.1752 -11.6547
27 C1b C 31.8841 -10.4279
28 O2a O 33.2500 -10.4285
29 C1b C 33.9499 -9.2175
30 C1a C 35.3498 -9.2181
31 C6a C 34.8376 -14.8105
32 C2b C 36.0495 -15.5131
33 C2b C 37.2671 -14.8105
34 C6a C 38.4790 -15.5131
35 O6a O 39.6908 -14.8105
36 O6a O 38.4790 -16.9123
37 O6a O 33.6257 -15.5131
38 O6a O 34.8376 -13.4113
BOND 38
1 11 12 1
2 9 13 1
3 12 14 1
4 10 11 1
5 13 15 1
6 9 10 1
7 14 15 1
8 13 16 1
9 12 17 1
10 17 18 1
11 18 19 1
12 19 20 1
13 20 21 1
14 17 21 2
15 19 22 2
16 22 23 1
17 23 24 2
18 24 25 1
19 20 25 2
20 18 26 1
21 26 27 1
22 27 28 1
23 28 29 1
24 29 30 1
25 1 2 1
26 1 3 1
27 1 4 2
28 2 5 2
29 5 6 1
30 6 7 1
31 6 8 2
32 31 32 1
33 31 37 1
34 31 38 2
35 32 33 2
36 33 34 1
37 34 35 1
38 34 36 2
BRACKET 1 32.2000 -17.4300 32.2000 -12.7400
1 40.8800 -12.7400 40.8800 -17.4300
1 2
ORIGINAL 1 1 2 5 6 7 8 3 4
REPEAT 1 31 32 33 34 35 36 37 38
|
|---|